Radioactive decay equations
Stability
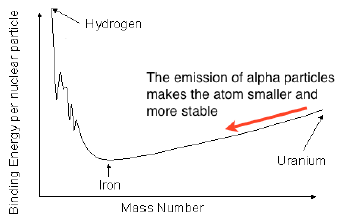
The most stable atoms are those around the size of iron with a mass number in the mid fifties. Large atoms can reduce their size, making them more stable by emitting alpha particles. Alpha particles have a mass of 4 amu having 2 protons and 2 neutrons. Small atoms can increase their size by fusion.
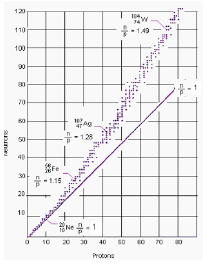
The balance of forces and energy within the nucleus is complex. There is an “ideal” ratio of neutrons to protons for isotopes, dependent on their size, that is shown by the irregular dotted band. The ratio within an atom is altered by beta or alpha decay. Beta decay increases the proportion of protons, beta plus decay and alpha decay reduce it. Electron capture neutralises a proton so increasing the proportion of neutrons.
Alpha decay
The loss of an alpha particle slightly decreases the p/n ratio (since there are more neutrons than protons in large atoms) as well as decreasing the size of the remaining nucleus.
The atomic mass reduces by 4 and the proton number by 2
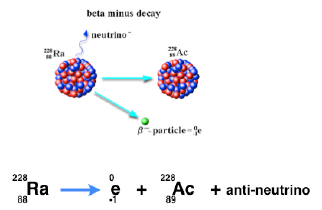
Beta minus decay
In beta minus decay a neutron breaks down into a proton and an electron. As a result the n/p ratio decreases.
The mass of the atom is not significantly changed (there is a small change due to the energy balance) but the proton number (atomic number) increases by 1.
In beta positive decay it is a proton that splits into a neutron and a positron. The energy required to produce a positron is significant and must be available from increasing the n/p ratio.
The mass of the atom is not
changed (there is a small change due to the energy balance) but the proton number (atomic number) decreases by 1.
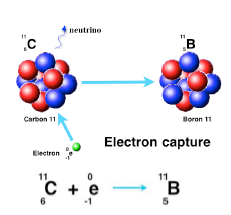
Electron capture
The capture of an electron into the nucleus which transforms a proton into a neutron increases the n/p ratio and requires less available energy than beta plus emission.
The proton number is one less but the mass is not significantly changed.
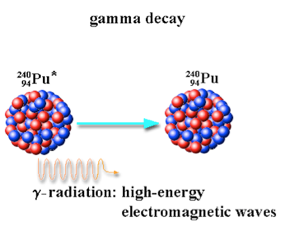
Gamma Radiation
Gamma rays are high energy short wavelength electromagnetic waves. Energy is often released this way at the same time or immediately after alpha or beta minus emission. Gamma radiation is also produced when a positron (beta plus particle) meets and annihilates an electron.
There are more useful pages about different aspects and uses of radioactivity here: